A
Turnkey Solution To Commercialize Your Healthcare products in the Asia Pacific Region.† We assist in
Registrations and
Business Development in the below mentioned Countries. We understand the legal requirements, cultural differences, geo-political situations in these countries and assist you in targeting the entire region via a
Single Trusted Partner. The product portfolio we currently handle include Medical Devices, IVDs, cosmetics, and Neutraceuticals.
Global manufacturers are showing immense interest in South East Asian countries including Philippines, Indonesia, Malaysia, Thailand etc (business friendly approach of the new government). Companies either start the process with market research, registrations and/or distributor search. We at Morulaa have a direct hands-on approach for all these processes. We believe that every project has its own requirements. It is important to have a personal direct contact with these medical device distributors, regulators, KOLs, hospital and other healthcare professionals.
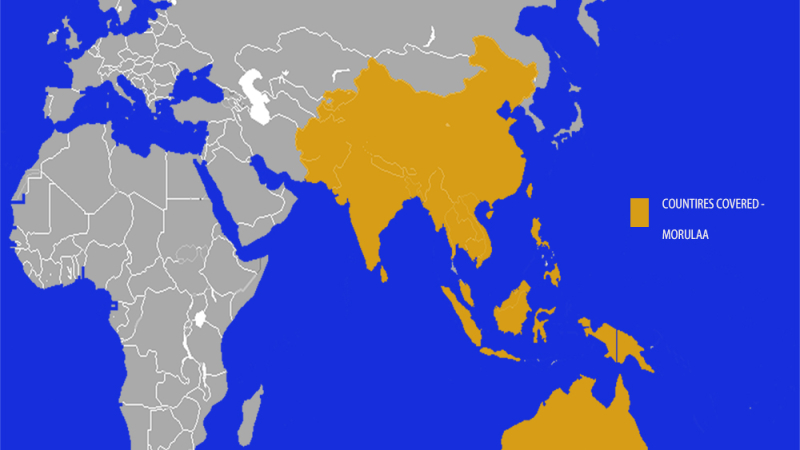
Below is the list of markets for which we provide Regulatory and Distribution Set-up Commercialize-ation services
Markets List the Commercialize-ation Services Provided:
To show that they are committed to encouraging foreign investments in the medical field, the governments are introducing a series of positive reforms including:
- Foreign equity caps in various sectors were relaxed or removed.
- Application of licenses made online
- Validity of licenses increased to 3 years
- Increase in foreign direct investments (FDI)
We have established collaborations with key companies across Asia Pacific and offer Regulatory and Commercialize-ation services across this region. Keep an eye on our blog to learn more about the regulatory and commercialize-ation service and requirements for various countries.