|
UDI: THE BARCODE THAT SAVES LIVES (AND YOUR SANITY)
|
|---|
|
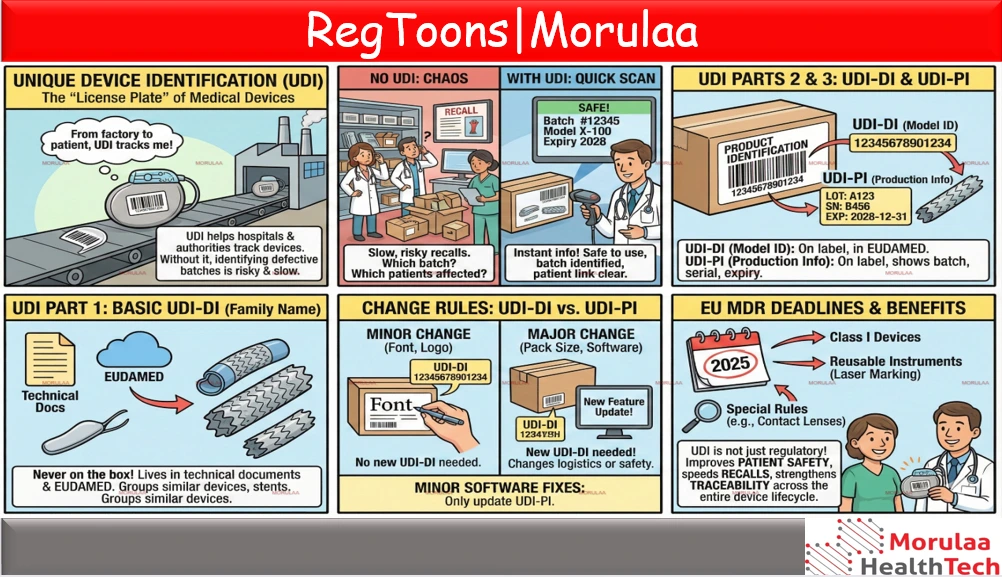
Unique Device Identification (UDI) is basically the "license plate" of a medical device. It helps hospitals and authorities know exactly which device is which from the moment it leaves the factory to the moment it reaches a patient. Without UDI, identifying defective batches is slow and risky. With UDI, a quick barcode scan instantly tells hospitals if a device is safe to use, which batch it came from, and which patients may be affected. UDI has three parts, and beginners often mix these up. Sometimes a change to the device requires a new UDI-DI but not always. Small updates like changing a font or logo do not require a new UDI-DI. But changes that affect logistics or safety such as creating a new pack size or releasing major new software functionality do require a new UDI-DI. Minor software fixes only need updated UDI production information. Finally, EU MDR has specific deadlines for when different devices must have UDI labels and, for reusable instruments, permanent laser marking. Most devices above Class I should already be compliant, while Class I devices and reusable instruments have deadlines in 2025. Some products like contact lenses have special rules and later dates. The main point is simple: UDI is not just a regulatory requirement; it improves patient safety, speeds up recalls, and strengthens traceability across the entire device lifecycle. |
THE "WHY": IT’S NOT JUST A STICKER
Imagine a hospital shelf stocked with 500 identical-looking boxes of hip implants. One specific batch has a manufacturing defect that causes fractures after 6 months.
- Without UDI: The hospital staff has to manually read every single box, checking product codes against a paper list. It takes weeks. Patients receive the bad implants during the delay.
- With UDI: The hospital ERP system scans a single barcode. It instantly identifies exactly which patient received the defective lot and tells the nurse, “Stop! Do not use this box,” before the package is even opened.
The Takeaway: UDI (Unique Device Identification) isn’t just paperwork. It is the “license plate” that allows a device to be tracked from our factory floor to the patient’s body.
THE ANATOMY OF A UDI (DON'T MIX THESE UP!)
The #1 cause of non-compliance is confusing the three layers of identification. Use this “Passport Analogy” to keep them straight.
Layer | The Analogy | What is it? | Where does it live? |
Basic UDI-DI | The “Family Name” | Identifies the group of devices with the same design/purpose. | Technical Docs & EUDAMED only. (NEVER put this on the box label!) |
UDI-DI | The “ID Number” | The static ID for a specific model (e.g., Catalog #1234). | On the Label + EUDAMED |
UDI-PI | The “Timestamp” | The dynamic production data (Lot #, Serial #, Expiry). | On the Label |
Visualizing the Carrier
The “Carrier” is the barcode itself.
- AIDC (Auto-ID): The barcode (GS1-128 or DataMatrix).
- HRI (Human Readable): The text numbers below the barcode (crucial if the scanner fails).
- DPM (Direct Part Mark): A laser-etched code on reusable steel instruments (more on this below).
THE "CHANGE" DECISION TREE: WHEN DO WE NEED A NEW ID?
One of the hardest parts of UDI is knowing when a change requires a new UDI-DI. Use this cheat sheet for your next Change Control meeting.Scenario A: The “Marketing” amendment
We updated the logo on the box and changed the font size of the instruction manual.
- Verdict: NO new UDI-DI.
- Reason: The device’s safety, performance, and identification remain unchanged.
Scenario B: The “Value Pack”
We currently sell Syringes in a box of 10. We are launching a new “Bulk Box” of 50.
- Verdict: YES, new UDI-DI.
- Reason: A change in package quantity creates a new “trade item” that must be distinguishable in inventory systems.
Scenario C: The Software Update
- Minor Update (v1.1 to v1.2): Bug fixes, faster loading. New UDI-PI only (the version number changes).
- Major Update (v1.0 to v2.0): We added a new diagnostic algorithm or changed the intended use. New UDI-DI required.
THE 2025/2026 DEADLINES: WHERE ARE WE NOW?
As of November 2025, we are in a critical compliance window.
For Standard Devices:
- Class III & Implantables: Past Due (2021) – Must be fully compliant.
- Class IIa / IIb: Past Due (2023) – Must be fully compliant.
- Class I (Low Risk): Deadline: May 26, 2025.
- Action Item: If we have any Class I devices (e.g., manual beds, simple tools) without UDI labels, they are now non-compliant.
For Reusable Surgical Instruments (The “DPM” Rule): - Class IIa/IIb Reusables: Deadline: May 26, 2025.
- The Challenge: These devices need “Direct Part Marking” (laser etching). The mark must survive hundreds of sterilization cycles.
- Pitfall: A common failure is buying cheap etching that fades after 50 autoclave cycles.
For Optical Devices (The “Master UDI-DI”):
- Context: Contact lenses have thousands of variants (diopters). Assigning a unique UDI-DI to each is impossible.
- Solution: The Master UDI-DI groups these variants.
- Contact Lenses: Mandatory by Nov 9, 2026.
- Spectacles/Frames: Mandatory by Sep 2028.
TEAM ACTION PLAN
To ensure we stay ahead, here is what every department needs to do:
- R&D: Ensure “UDI Assessment” is a mandatory checkbox in the Design Change form.
- Regulatory: Audit our Technical Files. Does every single SKU have a linked Basic UDI-DI?
- IT / Ops: Verify that our label printers are grading barcodes (ISO 15415 standards) to ensure readability.
- Quality: Check that our Declaration of Conformity lists the Basic UDI-DI (this is mandatory).
CONCLUSION: MAKE UDI WORK FOR YOU, NOT AGAINST YOU
UDI isn’t just a barcode or a compliance checkbox, it’s a smart tracking system that protects patients, speeds up recalls, and keeps your operations audit-ready. Whether you’re tweaking a label, launching a bulk pack, or rolling out software updates, understanding when to assign a new UDI is essential.
HOW MORULAA CAN HELP
Morulaa HealthTech supports medical device manufacturers in navigating the UDI requirements under EU MDR 2017/745 by offering end-to-end regulatory assistance. From assigning compliant Basic UDI-DIs and UDI-DIs through recognized issuing entities to ensuring accurate registration in EUDAMED, we help streamline the process. Our team integrates UDI implementation into your QMS, manages labeling and packaging reviews, and ensures your documentation aligns with EU regulatory expectations—minimizing compliance risks and accelerating market access.
