Overview of the PRRC Role under EU MDR and IVDR
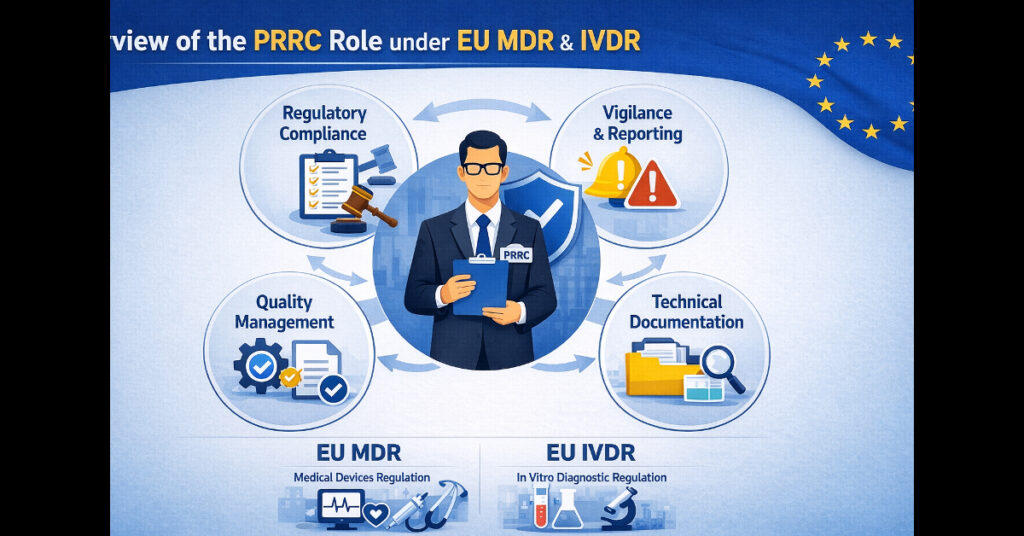
INTRODUCTION In the ever-evolving landscape of medical devices and in vitro diagnostic (IVD) regulations, compliance with the European Union Medical Device Regulation (MDR 2017/745) and the In Vitro Diagnostic Regulation (IVDR 2017/746) is paramount. A crucial role in ensuring that manufacturers comply with these regulations is that of the Person Responsible for Regulatory Compliance (PRRC). […]
AUSTRALIA’S TGA UPDATES ACCESS PATHWAY FOR UNAPPROVED MEDICAL DEVICES

TGA, December 29, 2025 — The Therapeutic Goods Administration (TGA) in Australia has released updated guidance on the Authorised Prescriber Scheme, clarifying how unapproved therapeutic goods including medical devices can be supplied legally before full registration. This update is a key development for device makers exploring early patient access in Australia. The TGA’s refreshed guidance […]
FDA Officially Unveils TEMPO Pilot: What Medical Device Manufacturers Must Know

December 2025 — Washington, D.C. — The U.S. The Food and Drug Administration (FDA) has formally announced the launch of the Technology‑Enabled Meaningful Patient Outcomes (TEMPO) for Digital Health Devices Pilot, marking a significant shift in how digital health technologies for chronic disease care may reach patients and markets. TEMPO is being introduced in coordination with […]
