TGA Amendment 2025: Australia Now Recognizes UK Certifications for Medical Devices

Australia 30, October 2025 — The Australian Therapeutic Goods Administration (TGA) has issued a pivotal update to its regulatory framework with the release of the “Therapeutic Goods (Medical Devices Information that Must Accompany Application for Inclusion) Amendment Determination 2025.” This amendment, effective 1 December 2025, introduces key changes that broaden the scope of acceptable documentation […]
General Safety and Performance Requirements (GSPRs) under IVDR 2017/746
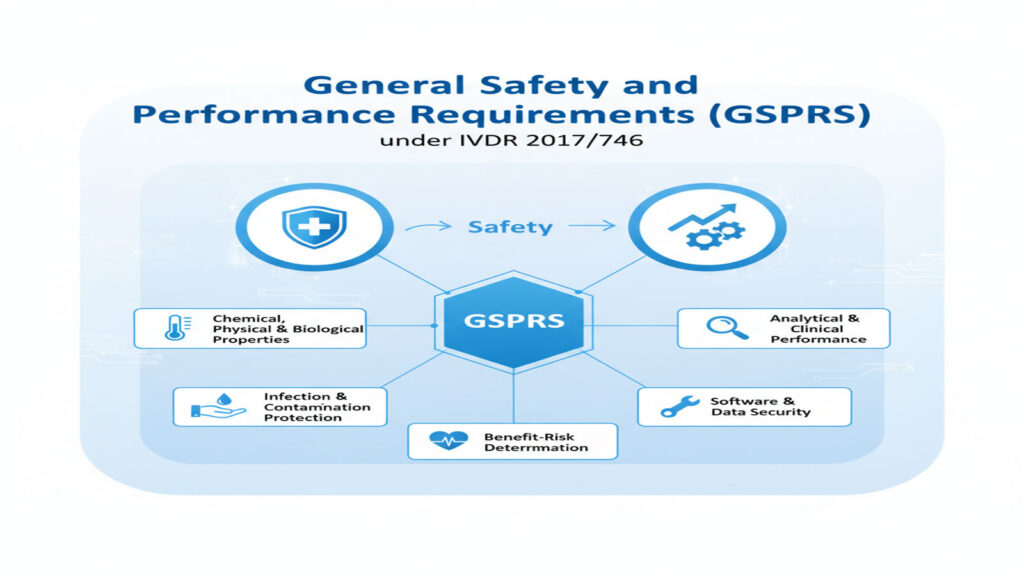
MEET THE GSPR: THE SAFETY RULES BEHIND EVERY IVD DEVICE The General Safety and Performance Requirements (GSPRs) in Annex I of the IVDR are the core rules that all IVD manufacturers must follow before placing a device on the EU market. They ensure that every device is safe, works as intended, and provides reliable results. […]
