Risk Management Report (RMR) under IVDR
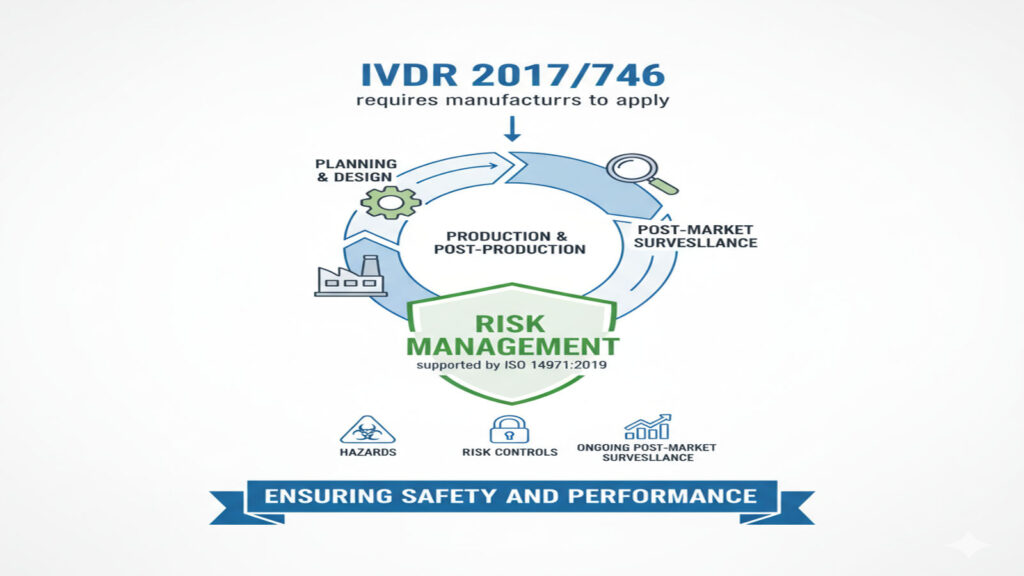
Keeping IVD Devices Safe: Risk Management Under IVDR Risk management is basically a structured way to make sure an IVD test is safe, works as intended, and stays that way over its whole life (design → manufacturing → use in the field). Under IVDR 2017/746, the manufacturer must set up and maintain a risk management […]
