Post-Market Surveillance (PMS) under IVDR 2017/746
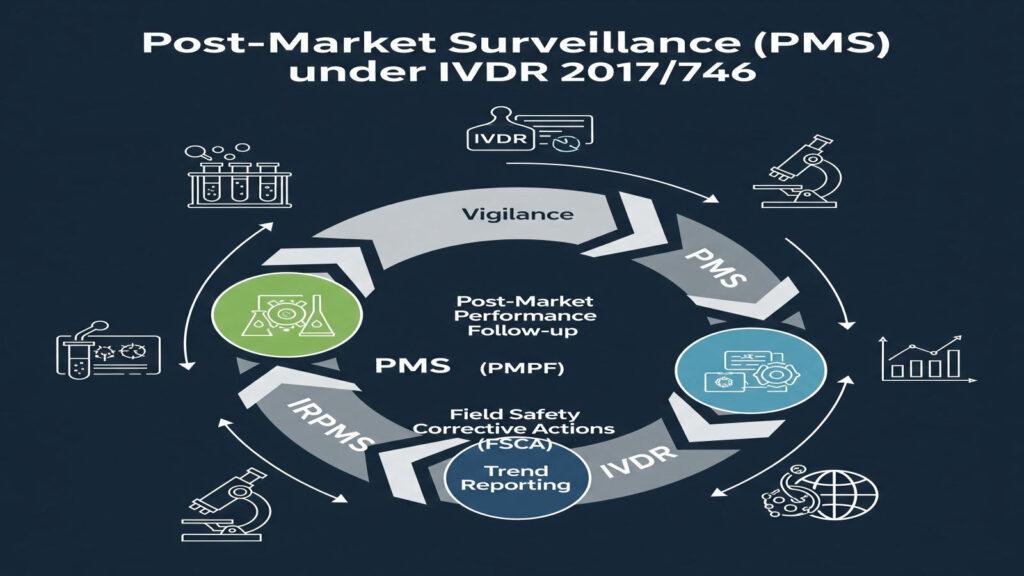
PMS Under IVDR: Keeping Your IVD Safe After Launch Post-Market Surveillance (PMS) means keeping watch on your IVD after it is sold and used in real life. PMS isn’t new (IVDD already expected manufacturers to collect post-production information), but IVDR 2017/746 makes PMS much more detailed and strictly enforced. The goal is simple: make sure […]
