Technical document under IVDR 2017/746
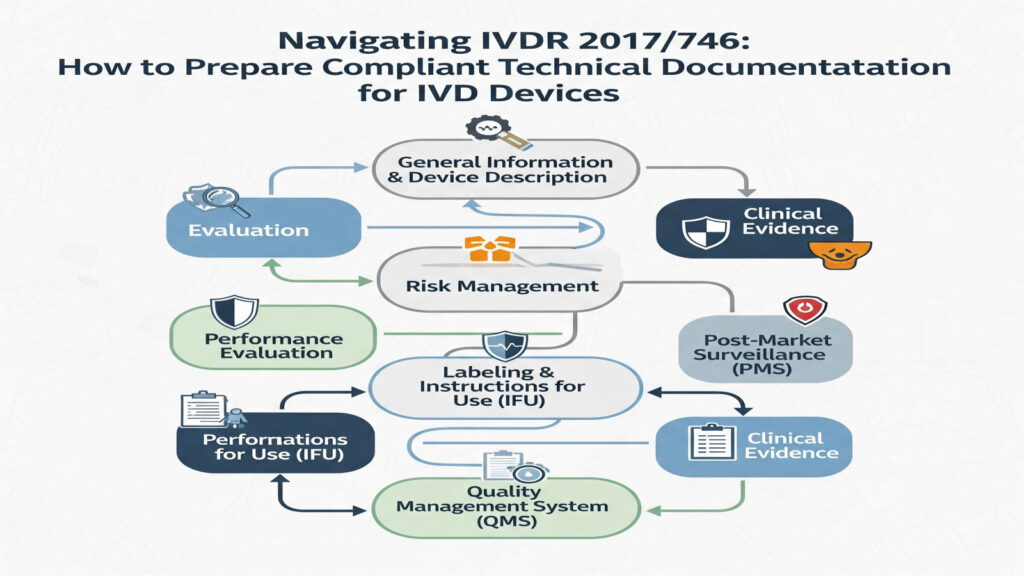
IVD Technical Documentation Made Simple IVD technical documentation is basically the master evidence file that proves your device meets IVDR requirements for safety, performance, and quality. Under IVDR 2017/746, you must prepare it before you place the device on the EU market and keep it updated for the whole life of the product because regulators […]
