Bans on Medical Devices by FDA Medical Device Regulations

Definition and Scope of FDA Device Bans A medical device ban is a complete restriction on all present and future production, distribution, and sales of medical devices if the FDA Medical Device Regulations believes that a medical device intended for human use offers a significant and unjustified risk of disease or harm, which cannot be […]
How to Report Medical Device Issues by FDA
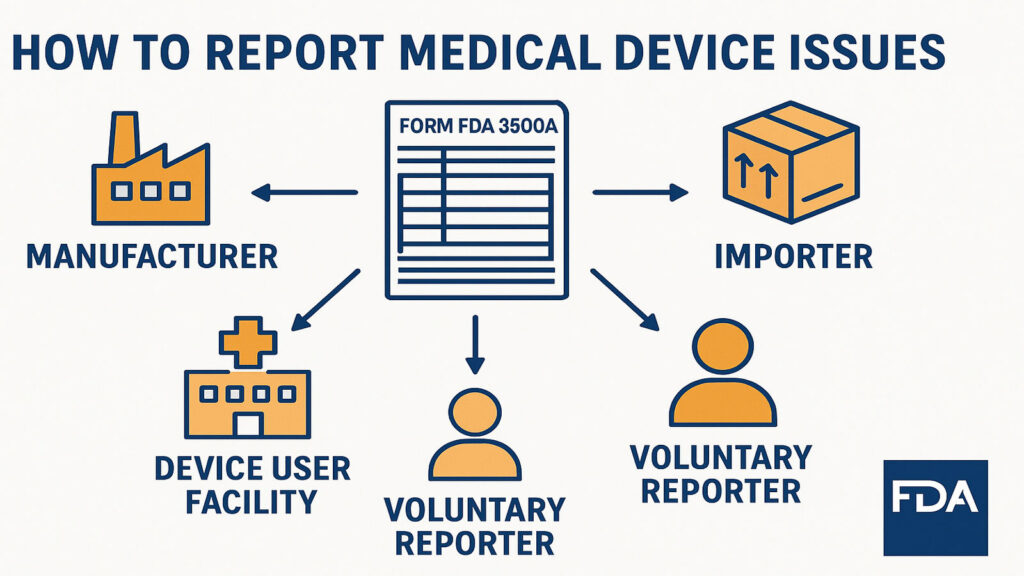
Understanding FDA Medical Device Reporting This article deals with mandatory reporters like manufacturers, importers, device user facilities and voluntary reporters who each submit medical device 510k reporting to the FDA. Medical device reports are a post market monitoring method used to track the functionality of devices, identify possible safety concerns, and contribute to benefit-risk analyses […]
